SRM Software for Food & Beverage:
Enforcing Food Safety & Ingredient Traceability
LeanLinking’s SRM Software for Food & Beverage centralizes supplier compliance, enforces food safety documentation, and maintains traceability across ingredients and packaging. Structured workflows ensure suppliers meet regulatory and safety requirements before materials enter production.

Purpose-Built SRM Software for Food & Beverage Supplier Governance
LeanLinking is a specialized SRM software platform designed for high-risk food and beverage supply chains. It centralizes supplier master data, governs food safety certifications such as GFSI, HACCP, and BRC standards, and enforces structured traceability across ingredients, packaging, and other production-critical inputs.
By connecting quality teams, procurement, and supply chain operations within a single governance platform, LeanLinking ensures supplier compliance documentation, deviation management, and ingredient traceability are validated before materials enter production, reducing contamination risk and preventing costly product recalls.
Centralized Food Safety Documentation
LeanLinking centralizes supplier master data and food safety certifications such as GFSI, HACCP, and BRC standards within a controlled system of record, ensuring manufacturers always operate with validated supplier compliance documentation.
Enforced Supplier Deviation Management
Structured quality workflows ensure supplier deviations are documented, investigated, and resolved through controlled processes before ingredients, packaging, or other materials are approved for use in production.
Ingredient and Packaging Traceability
LeanLinking maintains complete traceability across ingredients, packaging materials, and supplier documentation. This enables manufacturers to quickly identify contamination risk, support audit readiness, and prevent recall exposure.
The Failure of Manual Management: Expired Certificates and Recall Risks
Many food and beverage producers still manage supplier compliance through Excel trackers and email attachments. While convenient, this fragmented approach creates critical compliance blind spots in supply chains where contamination tolerance is zero.
When certifications, ingredient traceability documentation, and supplier deviations are tracked across spreadsheets and inboxes, organizations lose visibility into food safety compliance and supplier risk.

Expired Food Safety Certificates
Food safety certifications (BRC, ISO 22000, and GFSI standards) often expire unnoticed when managed through inboxes and spreadsheets, allowing suppliers to remain active without validated compliance documentation.

Broken Ingredient Traceability
Ingredient and packaging traceability records frequently remain scattered across departments and attachments, preventing food manufacturers from maintaining a reliable contamination traceability history.
Untracked Supplier Deviations
When supplier quality deviations are logged informally through email communication and spreadsheets, investigation documentation and corrective action tracking become inconsistent and difficult to audit.

Recall and Regulatory Risk
Without structured supplier compliance governance, expired certifications and unresolved quality deviations increase the likelihood of failed audits, contaminated batches, product recalls, regulatory penalties, and brand damage.
Core Pillars of Food & Beverage Supplier Governance
Effective food safety governance requires structured control across supplier qualification, deviation resolution, and performance oversight.
Pre-Qualification and Automated Food & Beverage Safety Compliance
Effective food and beverage safety governance starts at the point of qualification, before any ingredients, packaging, or other production-critical inputs are approved for use in production.
LeanLinking mandates structured supplier onboarding workflows that require submission of food and beverage safety documentation (e.g., GFSI-recognized certificates, HACCP plans, allergen management documentation) prior to supplier approval and activation.
This enforcement model eliminates non-compliant suppliers at the point of entry by deploying an automated Supplier Onboarding Software that strictly enforces a “No Document, No PO” standard.
LeanLinking continuously monitors certificate expiry dates, automatically issuing renewal notifications before compliance gaps emerge. If required documentation lapses, the system flags the issue immediately and restricts supplier approval status until valid documentation is restored.
This structured control ensures food producers retain full oversight of which suppliers are authorized to provide ingredients, packaging, or other production-critical inputs into the manufacturing process.
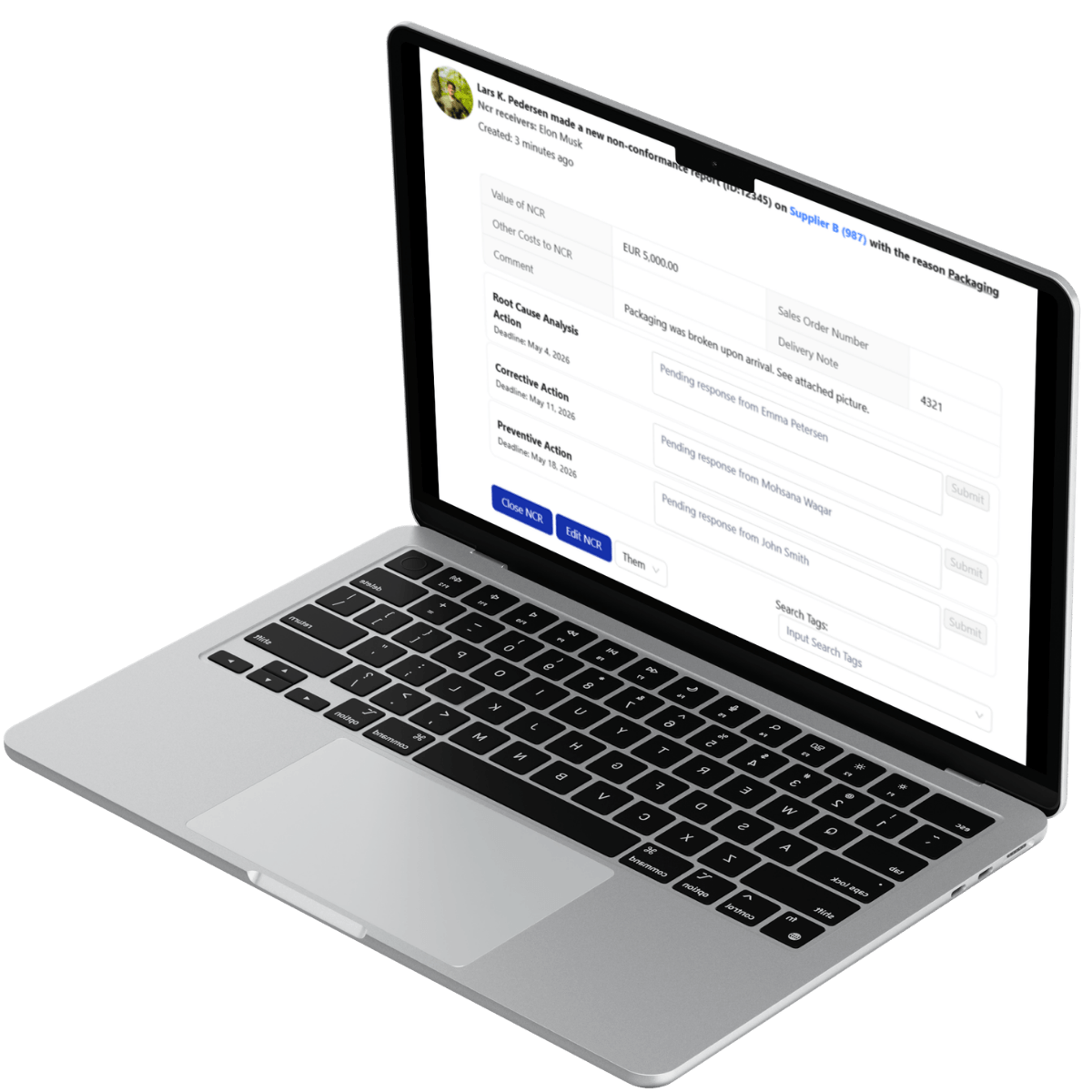
Deviation Governance: NCRs and Recall Prevention
Across food and beverage supply chains, every deviation must be traceable to a specific batch, production run, and supplier.
LeanLinking enforces structured Supplier Quality Management workflows that trace deviations (e.g., NCR) back to the precise supplier and production lot. Suppliers are unable to close a deviation case without submitting documented root cause investigation and preventive action evidence (e.g., RCA reports and corrective action documentation).
LeanLinking ensures contamination events are investigated systematically, and corrective measures are validated before continued supply is authorized.
Scandi Standard implemented LeanLinking’s NCR and compliance workflows across Quality, Procurement, and Supply Chain functions, replacing legacy Excel-based tracking with structured process mapping and controlled approval logic. The result was significant time savings in supplier documentation collection, improved cross-functional data sharing, and continuous audit readiness across factory operations.
By formalizing deviation traceability and corrective action enforcement with LeanLinking, food and beverage producers reduce recall exposure and protect consumer safety.
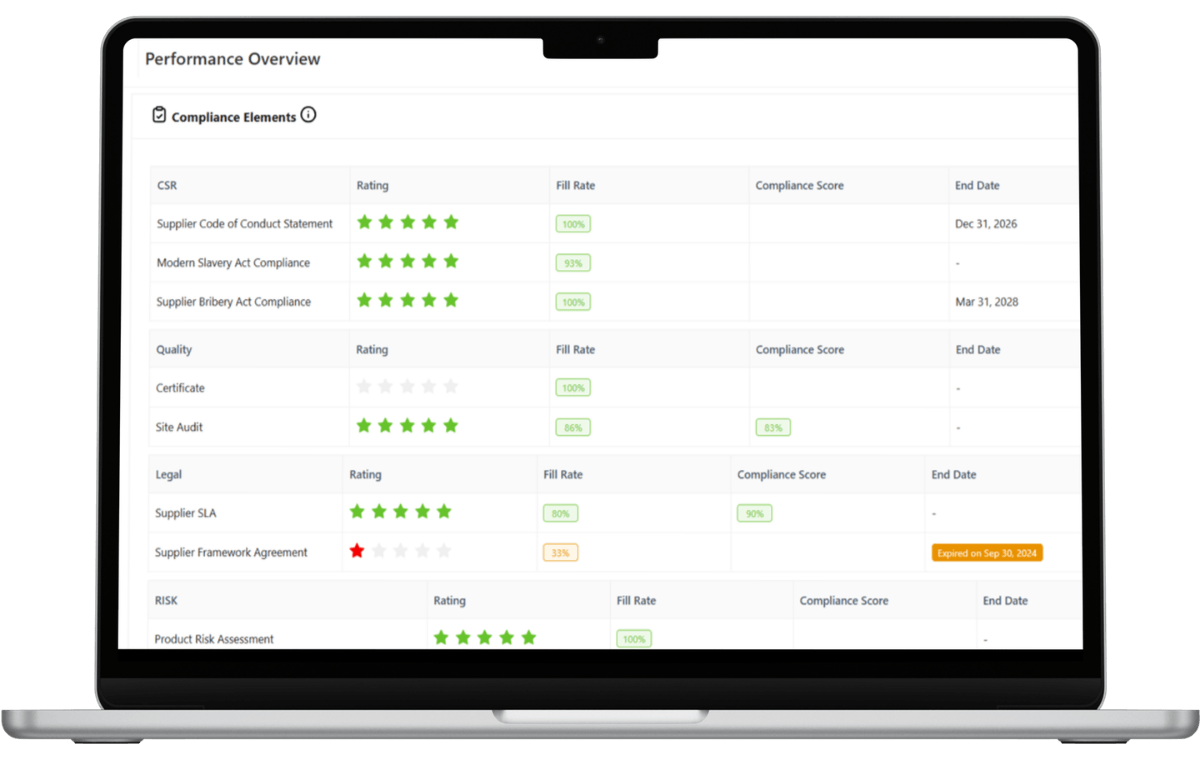
Digital Supplier Audits and Objective Performance Scorecards
Annual food safety audits alone do not provide continuous oversight across complex supply networks. LeanLinking digitizes supplier audits (e.g., food safety self-assessments and hygiene compliance reviews), tracks responses in real time, and automatically triggers follow-up workflows when results fall below defined safety thresholds. This transforms periodic audits into continuous supplier risk monitoring.
LeanLinking also consolidates delivery reliability metrics such as OTIF with documented compliance history to generate structured Supplier Performance Scorecards, providing a unified view of supplier performance and food safety adherence.
Good Food Group deployed LeanLinking to centralize compliance, performance, and quality management across more than 700 Tier 1 suppliers and over 300 users. The platform consolidated supplier documentation, automated non-conformance case management, and integrated ERP performance data such as OTIF delivery metrics into a single governance environment. This reduced audit preparation time from over a month to a few days and improved supplier administrative efficiency by more than 50 percent.
With LeanLinking, supplier performance oversight becomes structured, measurable, and defensible.
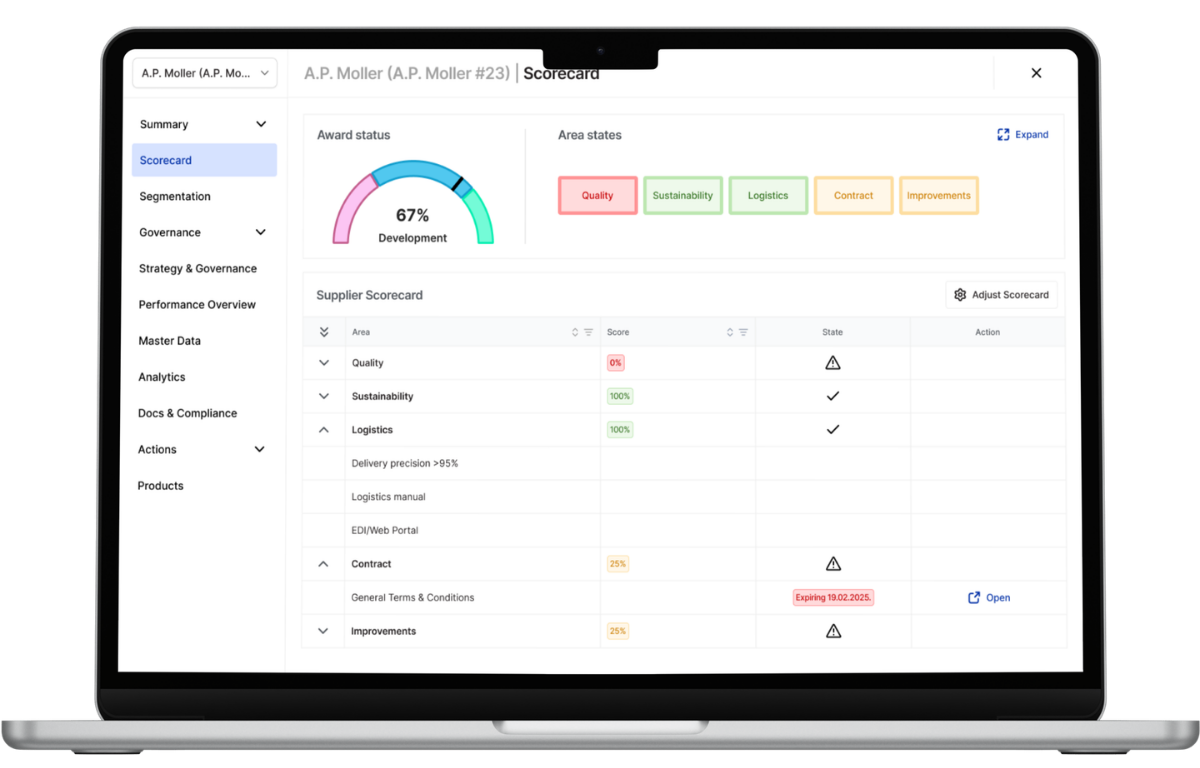
Transition to the Operational Supplier Cockpit for Food Safety Governance
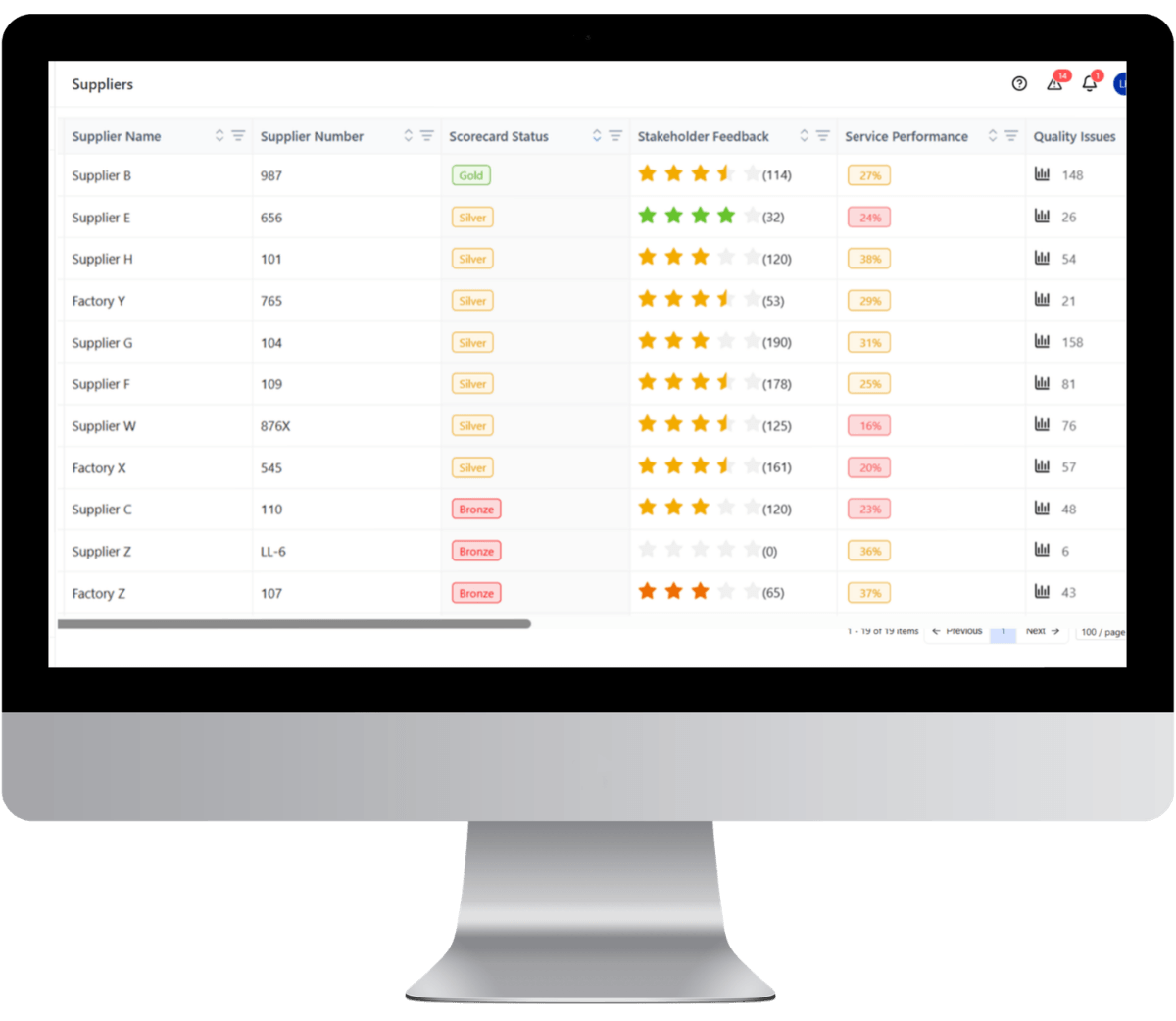
Maintaining zero-contamination tolerance across complex food and beverage supply chains requires replacing fragmented spreadsheets and email-based tracking with a centralized Supplier Cockpit. LeanLinking provides a unified environment where supplier compliance documentation, quality deviations, and delivery performance are monitored continuously.
Centralized Food Safety Compliance
LeanLinking consolidates supplier food safety documentation into a structured compliance framework so manufacturers maintain continuous visibility over supplier certification and regulatory requirements.
GFSI certification documentation
HACCP compliance records
BRC audit documentation
Continuous certification monitoring
Traceable Quality Incident Management
Deviation and recall-related quality incidents are managed through structured workflows that ensure every case remains traceable from detection through resolution.
Non-conformance reports (NCR)
Root cause investigations
Corrective action documentation
Complete deviation traceability
Supplier Risk and Performance Visibility
LeanLinking combines delivery performance monitoring with compliance and deviation history to provide a complete view of supplier risk across the supply network.
OTIF delivery reliability monitoring
Ingredient and packaging traceability
Automated compliance expiry alerts
Real-time supplier risk visibility
Testimonials

"LeanLinking is a game changer for working with suppliers. For the first time we can close the supplier management cycle from strategy through governance to performance managements and supplier development."

"Our team loves easy to use interface; with Relations, we have managed to cut non-compliance resolution time by half, and migrated interactions with our vendors almost fully to LeanLinking."

Strengthen Food Safety Governance Across Your Supply Chain
LeanLinking centralizes supplier food safety documentation, enforces structured deviation resolution, and maintains full traceability across ingredients and packaging. Replace fragmented spreadsheets with enforceable supplier governance designed for regulated food and beverage supply chains.
Food & Beverage Supplier Management FAQs
Why can’t ERP systems prevent food safety contamination risks?
Enterprise Resource Planning systems (e.g., SAP, Microsoft Dynamics) process purchase orders and manage inventory levels. They are Systems of Record designed for financial and operational transactions. However, they do not enforce food and beverage safety governance at the root cause level.
An ERP confirms that ingredients were received, but it does not validate whether the supplier’s food and beverage safety certification (e.g., GFSI-recognized audit, HACCP documentation) was valid at the time of delivery. It does not mandate structured root cause investigation when contamination or packaging failures occur. Additionally, an ERP does not maintain a complete traceable audit trail across supplier deviations.
LeanLinking operates above the ERP as an active System of Engagement that validates food and beverage safety standards and mandates corrective actions before ingredients, packaging, and other production-critical inputs enter the production line.
When a quality deviation is detected:
- LeanLinking generates a structured deviation record (e.g., NCR)
- The supplier must submit documented root cause investigation and corrective action documentation (e.g., RCA reports and preventive action plans)
- Defined approval workflows validate corrective action completion
- Each action generates a time-stamped, traceable record
LeanLinking does not function as a transactional system. It enforces structured supplier accountability before contaminated ingredients, packaging, or other production-critical inputs reach the manufacturing environment. In perishable supply chains, risk control must begin before goods are released into production.
Real Stories, Real Results
E-Book · 4 min
Case Study · 4 min




