SRM Software for Pharma: Enabling GxP Supplier Compliance and Audit Readiness
LeanLinking’s SRM Software for Pharma is a centralized operational governance platform that automates the lifecycle of compliance documentation (e.g., GMP certificates, FDA audit reports), strictly enforces deviation resolution (e.g., CAPA, NCR workflows), and guarantees continuous GxP audit readiness across complex pharmaceutical supply chains.

Purpose-Built SRM Software for Pharmaceutical Supplier Compliance
LeanLinking is a specialized SRM software (internal linking to What is SRM) for pharmaceuticals that centralizes supplier master data, automates the lifecycle management of regulatory documentation (e.g., GMP certifications and ISO standards), and enforces structured audit trails to support organizations operating under Good Practice regulations (GxP).
Centralized Supplier Compliance Data
LeanLinking centralizes supplier master data and regulatory documentation such as GMP certificates and ISO standards in a controlled system of record.
Automated Documentation Lifecycle Control
Supplier documentation is monitored throughout its lifecycle. Expiry tracking and automated alerts ensure regulatory certifications remain valid before APIs, excipients, or packaging materials enter production.
Structured Audit Trails for GxP Compliance
Documented workflows generate traceable audit records for supplier approvals, deviation resolution, and compliance verification under GMP and broader GxP frameworks.
The Failure of Manual Management: Excel Silos and FDA Audit Risks
Many pharmaceutical organizations still manage supplier compliance through Excel spreadsheets and Outlook email threads. While flexible, this decentralized approach creates serious regulatory risk. Critical compliance documentation, deviation records, and corrective action evidence often remain scattered across inboxes and spreadsheets rather than controlled systems.
Manual processes introduce compliance blind spots that weaken audit readiness and regulatory defensibility.

Uncontrolled Compliance Documentation
Regulatory certifications (e.g., GMP documentation, quality certificates, FDA registrations) often expire unnoticed in individual inboxes, leaving organizations unknowingly operating out of compliance.
Fragmented Deviation Investigations
Deviation investigations are handled through informal communication and email attachments, resulting in fragmented resolution documentation and incomplete audit history.

Broken Compliance Traceability
When documentation is distributed across attachments and personal folders, organizations lose the structured audit trail required to demonstrate supplier oversight during inspections

Regulatory and Operational Risk
These blind spots increase the likelihood of inspection findings, warning letters, product recalls, and production delays caused by incomplete supplier compliance documentation.
Core Pillars of Pharmaceutical Supplier Governance
Pharmaceutical supplier governance requires disciplined control across documentation validity, deviation resolution, and performance oversight.
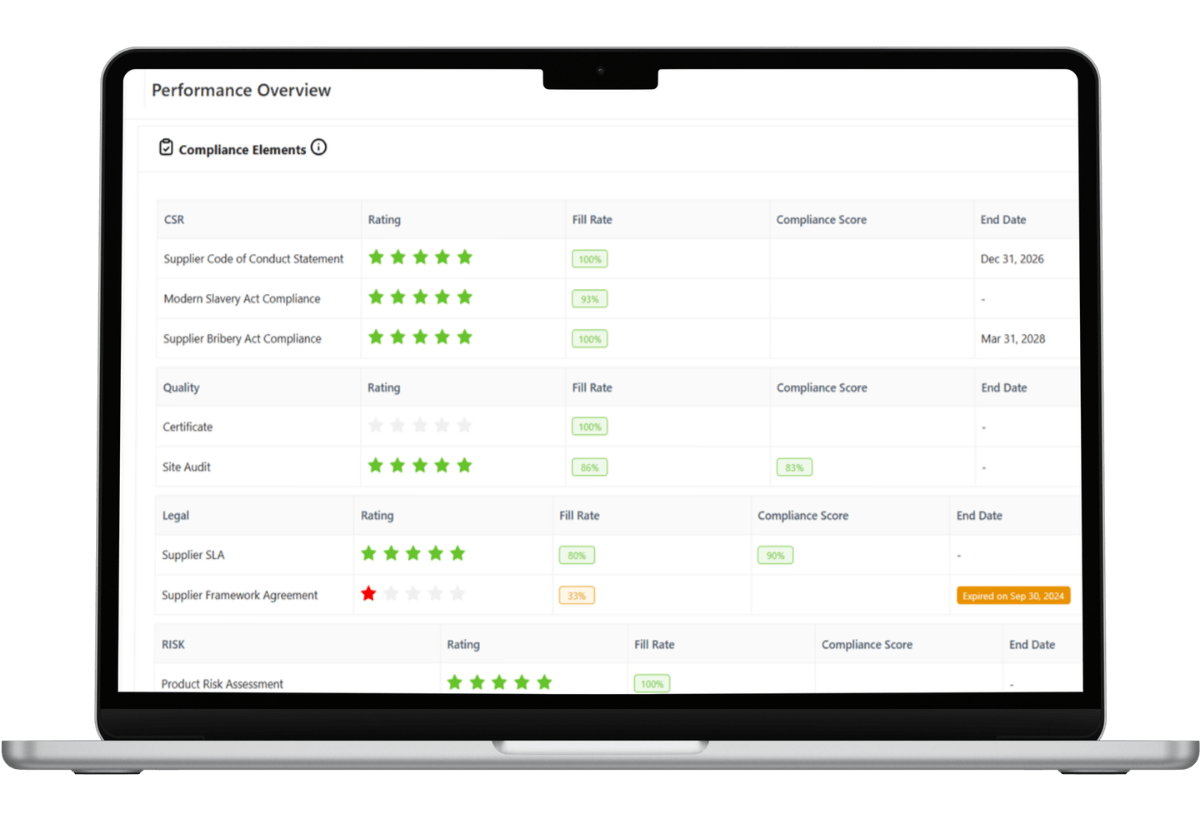
Automated Certificate Management and Audit Trails
Supplier compliance begins with valid documentation.
LeanLinking centralizes regulatory documentation (e.g., GMP certificates, ISO-based quality certifications, FDA documentation, standard operating procedures) within a
structured compliance archive. Expiry dates are monitored automatically, and real-time alerts flag upcoming certificate lapses before they create compliance exposure. Suppliers cannot remain approved if required documentation is missing or expired.
Audit readiness becomes continuous rather than reactive.
During inspections, Quality Assurance teams access validated supplier documentation instantly. Certificate histories, approval records, renewal timelines, and documented deviation logs are maintained within a centralized, traceable system of record.
LeanLinking enforces the documentation controls and traceability mechanisms that enable pharmaceutical organizations to demonstrate that their suppliers meet required GMP and broader GxP standards.
With LeanLinking documentation is not stored passively. Validity is continuously monitored, and compliance evidence remains structured, accessible, and defensible.
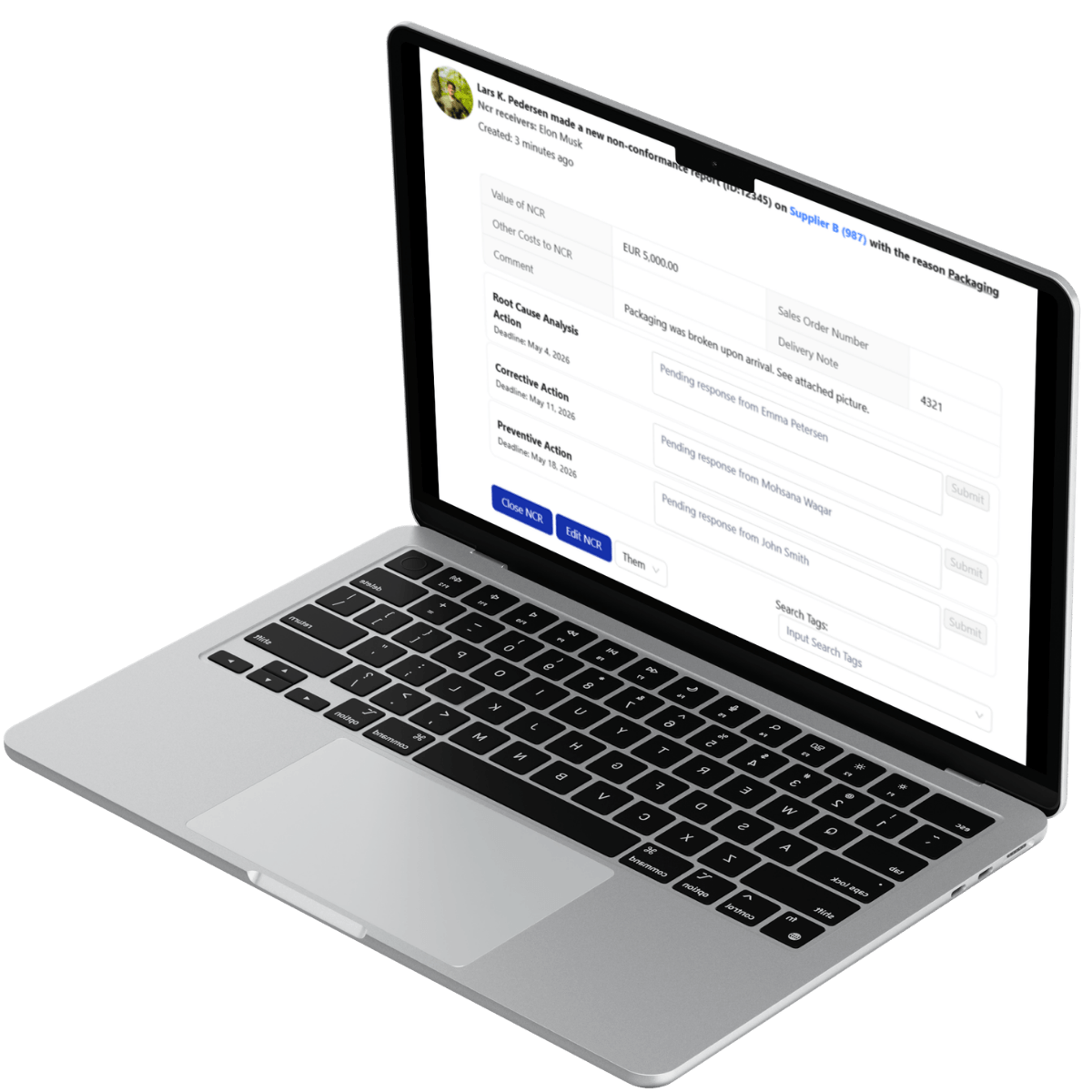
Deviation and Corrective Action Lifecycle Enforcement
In pharmaceutical supply chains, deviations must be documented, investigated, and resolved systematically.
LeanLinking digitizes structured
deviation management workflows (e.g., NCRs) and mandates documented root cause investigation and remediation processes (e.g., RCA documentation and CAPA plans).
A deviation case remains open until documented remediation evidence is reviewed, validated, and formally approved. Closure without corrective documentation is blocked by defined workflow controls.
Every approval action generates a permanent audit entry.
This enforcement structure ensures that supplier deviations are not resolved informally. Each case produces documented evidence aligned with GMP expectations for traceability and corrective action control.
LeanLinking enables pharmaceutical organizations to demonstrate that deviations were investigated, corrective actions were implemented, and supplier accountability was enforced.
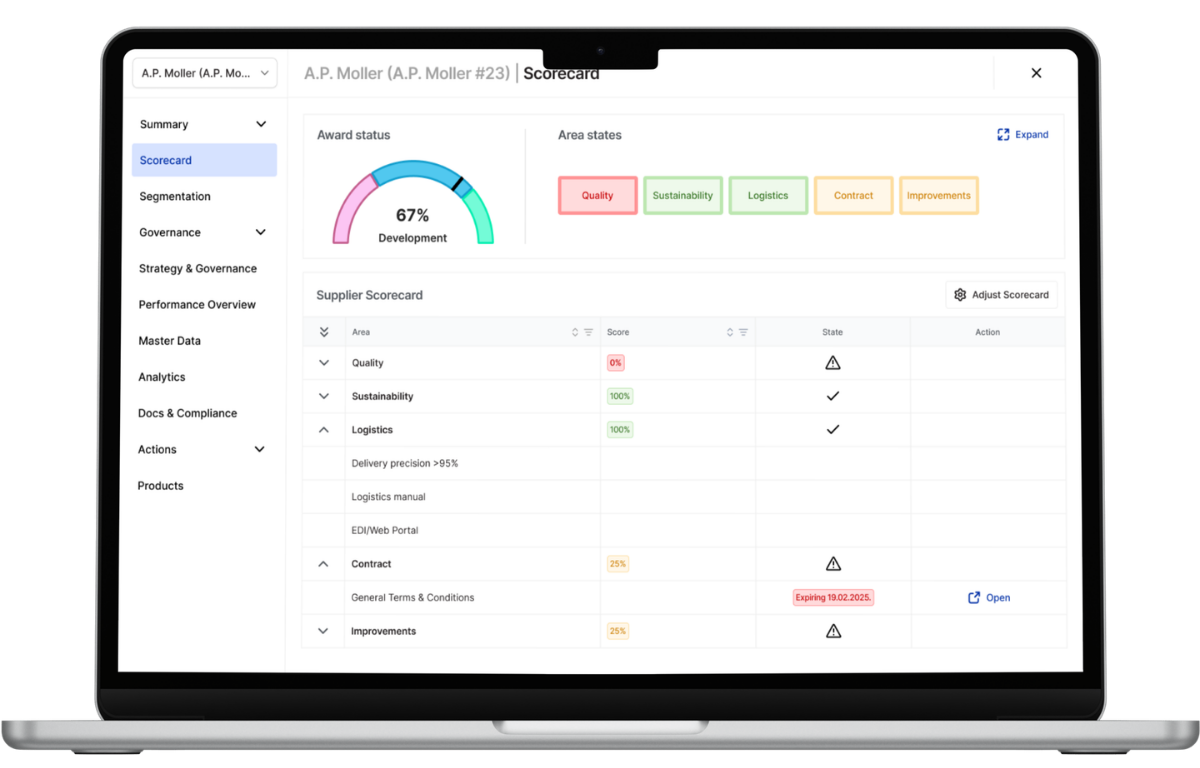
Pre-Qualification and Objective Performance Scorecards
Supplier compliance oversight begins before first shipment.
LeanLinking enforces structured
supplier onboarding workflows that require supplier to submit regulatory documentation (e.g., GMP certifications, quality standards, controlled process declarations) before supplier approval and activation. Suppliers self-validate required information through controlled forms, reducing manual data entry errors.
Roche collaborated with LeanLinking to centralize supplier data, monitor performance, and automate compliance documentation validation across complex pharmaceutical supply chains. This structured oversight reduced compliance blind spots and strengthened audit transparency across global supplier networks.
Post-qualification, LeanLinking consolidates measurable delivery performance indicators (e.g., OTIF adherence) with structured compliance metrics to generate risk-based supplier segmentation.
With LeanLinking Quality Assurance and Procurement leaders gain objective visibility into which suppliers maintain strong documentation discipline and operational reliability. Compliance oversight becomes more measurable than assumed.
Transition to the Operational Supplier Cockpit
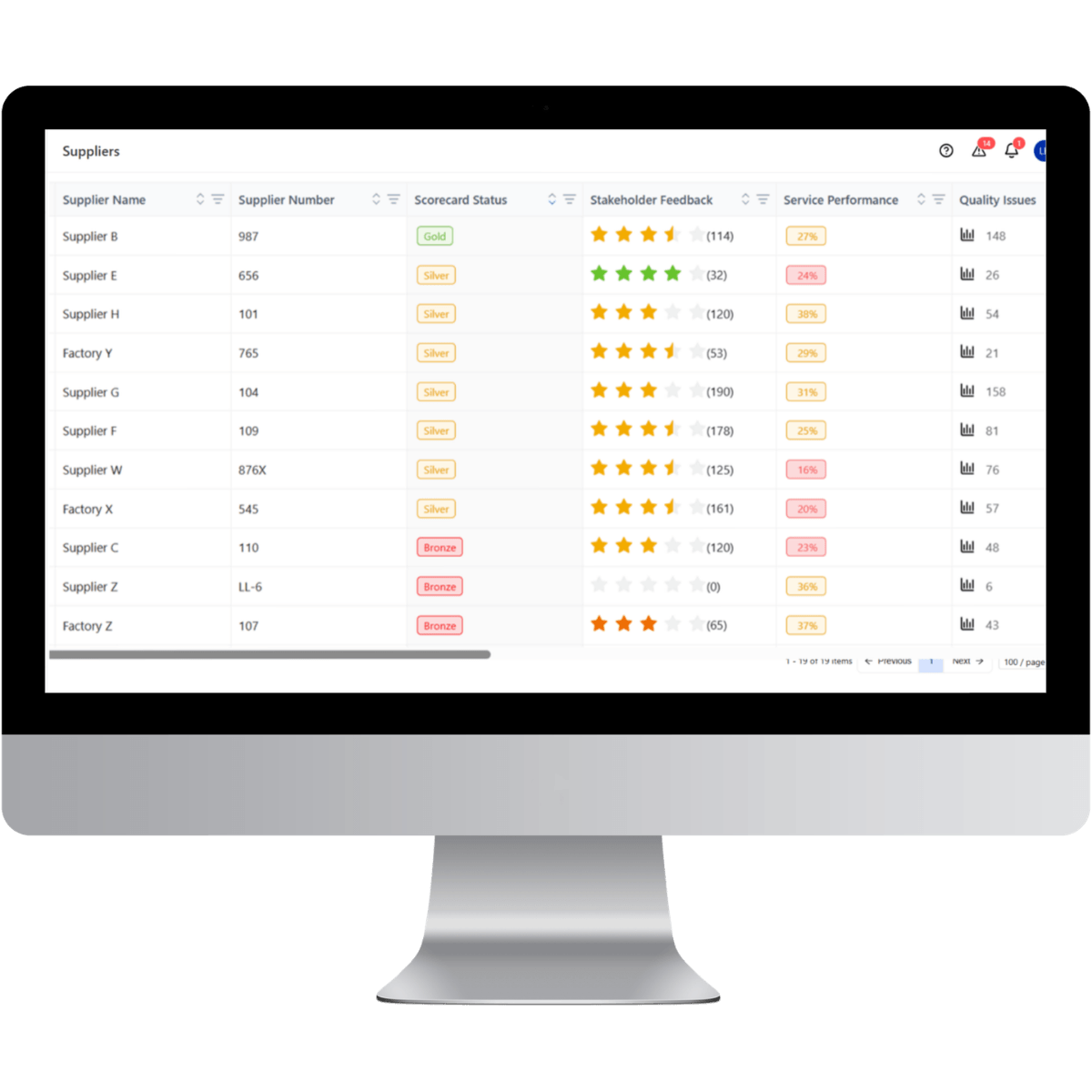
Pharmaceutical organizations cannot maintain structured supplier compliance using spreadsheets and email-based deviation tracking. LeanLinking replaces fragmented oversight with a centralized Supplier Cockpit that continuously enforces documentation control, deviation management, and audit-ready traceability across pharmaceutical supply chains.
Centralized Compliance Control
LeanLinking centralizes regulatory documentation and supplier compliance records within a controlled system of record, ensuring pharmaceutical organizations maintain continuous visibility over supplier qualification.
- GMP certificates
- ISO standards
- FDA documentation
- Compliance declarations
Structured Deviation Governance
LeanLinking enforces structured deviation management workflows that require documented investigation and validated remediation before issues are closed.
- NCR deviation records
- Root cause investigations (RCA)
- Corrective and preventive actions (CAPA)
- Controlled deviation resolution workflows
Continuous Audit-Ready Oversight
LeanLinking connects Quality Assurance, Regulatory Affairs, and Procurement teams within a unified supplier governance framework that ensures compliance obligations remain documented and traceable.
- Real-time compliance monitoring
- Supplier performance analytics
- OTIF delivery reliability metrics
- Complete audit trail generation
Testimonials

"LeanLinking is a game changer for working with suppliers. For the first time we can close the supplier management cycle from strategy through governance to performance managements and supplier development."

"Our team loves easy to use interface; with Relations, we have managed to cut non-compliance resolution time by half, and migrated interactions with our vendors almost fully to LeanLinking."

Ensure Supplier Compliance Across Your Pharmaceutical Supply Chain
LeanLinking provides the structured workflows, documentation controls, and traceability required to demonstrate supplier compliance under GMP and broader GxP regulations.
Pharmaceutical Compliance FAQs
What is the difference between Pharma SRM and ERP systems for GMP compliance?
Enterprise Resource Planning systems (e.g., SAP, Oracle) are designed to process purchase orders and record invoices. They are transactional engines, not supplier compliance governance systems.
An ERP confirms that materials were received, but it does not validate whether the supplier’s Good Manufacturing Practice certification (GMP) was current at the time of shipment. It also lacks the ability to mandate documented root cause investigation when deviations occur or preserve structured audit evidence of corrective action approval. While transactional systems record activity, they do not enforce supplier compliance governance.
LeanLinking operates above the ERP as an active System of Engagement designed specifically for pharmaceutical supplier governance.
When a supplier deviation occurs:
- LeanLinking issues a structured deviation record (e.g., NCR)
- The supplier must submit documented root cause investigation and remediation evidence (e.g., RCA reports & CAPA plans)
- Approval workflows validate corrective action completion
- Every step generates a time-stamped, traceable audit record
LeanLinking enforces processes and documentation controls that allow pharmaceutical organizations to demonstrate supplier compliance under GMP and broader GxP frameworks.
LeanLinking ensures that documentation becomes structured, complete, and traceable evidence.
Real Stories, Real Results
E-Book · 4 min
Case Study · 4 min




