Supplier Management Software:
The Operational Cockpit for Regulated Industries
LeanLinking’s Supplier Management Software centralizes operational governance system that automates supplier documentation lifecycle management (certificates, safety sheet, code of conducts etc.), enforces Non-Conformance (NCR) workflows, and aggregates supplier performance data to mitigate supply chain risk and maximize value creation in the supply chain.
Structured Governance for Supplier Quality Management
LeanLinking’s Supplier Quality Management Software is a collaborative governance system that digitizes Non-Conformance Reports (NCRs), mandates Root Cause Analysis (RCA), enforces Supplier Corrective Action Requests (SCARs), and centralizes supplier audit data to prevent quality failures from reaching production. It functions as an ongoing enforcement engine in regulated supply chains where unresolved deviations result in operational disruption, regulatory exposure, and commercial loss.
Structured Quality Issue Management
LeanLinking digitizes supplier quality workflows by capturing defects, enforcing root cause investigation, and managing corrective actions within a centralized governance system.
Automated Quality Enforcement
The platform acts as an automated quality firewall that blocks unresolved defects, validates corrective actions before closure, and ensures issues cannot be informally resolved or ignored.
Continuous Compliance Governance
Quality governance cannot be periodic. LeanLinking ensures compliance oversight is continuous, documented, and enforceable across the supplier base.
The Failure of Manual Management: Excel Silos and Compliance Blind Spots
Manual supplier management via Excel spreadsheets and Outlook creates data silos, leaving procurement teams unaware of expired certificates, decreasing supplier performance until an audit failure occurs and there is a serious delivery issue.
Three structural risks define manual oversight:

Data Silos
Certificates, performance metrics, documentation and NCR logs exist in disconnected files.

Tribal Knowledge
Critical supplier data resides in one buyer’s inbox, creating a single point of failure.

Reactive Firefighting
Teams respond after disruption instead of preventing it.
Automated Compliance, Master Data, Performance and NCR Workflows
Modern Supplier Management Software govern three core operational clusters.
Automated Certificate Lifecycle Management (Certificates eg. ISO 14001 & Eco-Labels)
LeanLinking operates as a compliance shield.
The platform automates tracking of environmental and ethical certifications, including:
- ISO 14001
- FSC
- Fair Trade
- Supplier Codes of Conduct
When certifications approach expiration, LeanLinking sends automated reminders to suppliers. If renewal documentation is not uploaded, escalation workflows can activate.
Good Food Group leverages LeanLinking to maintain strict sustainability and safety documentation compliance across its ingredient supply chain. Documentation remains current and audit-ready.
This is structured Automated ESG compliance that validates certificates before exposure occurs.
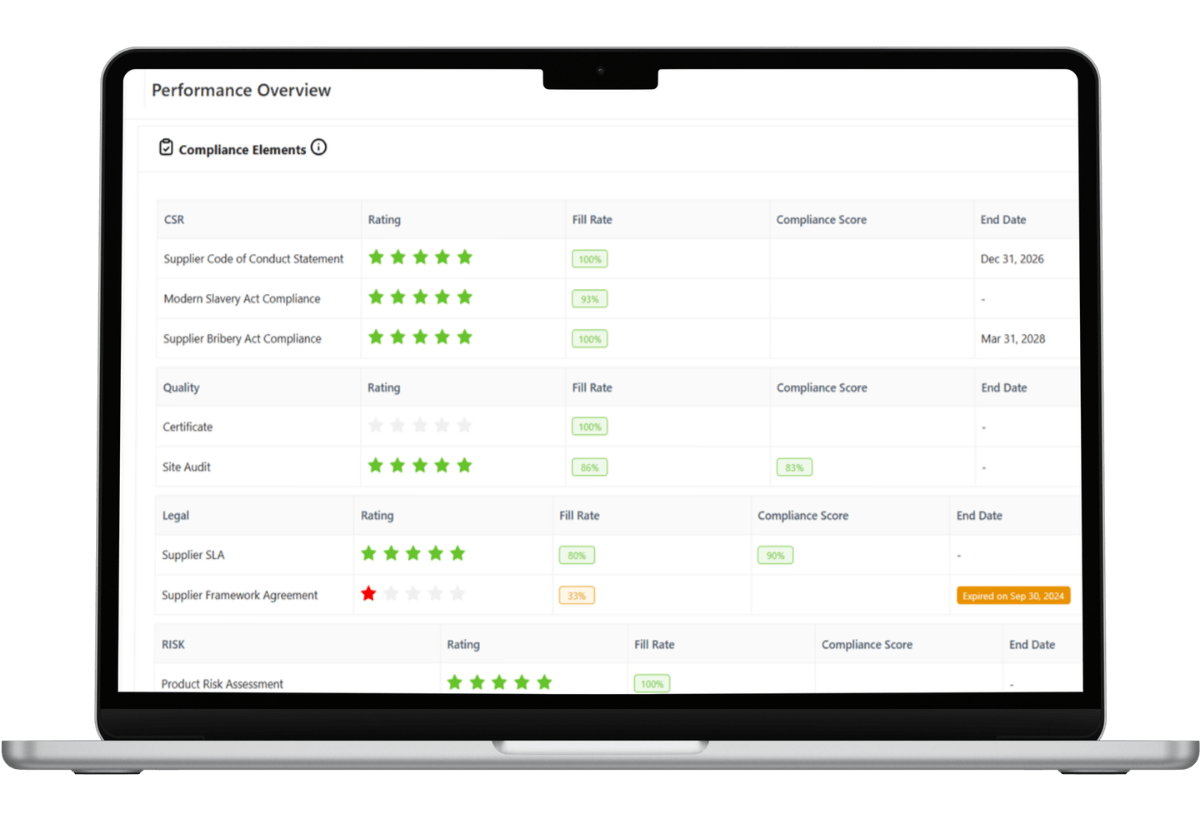
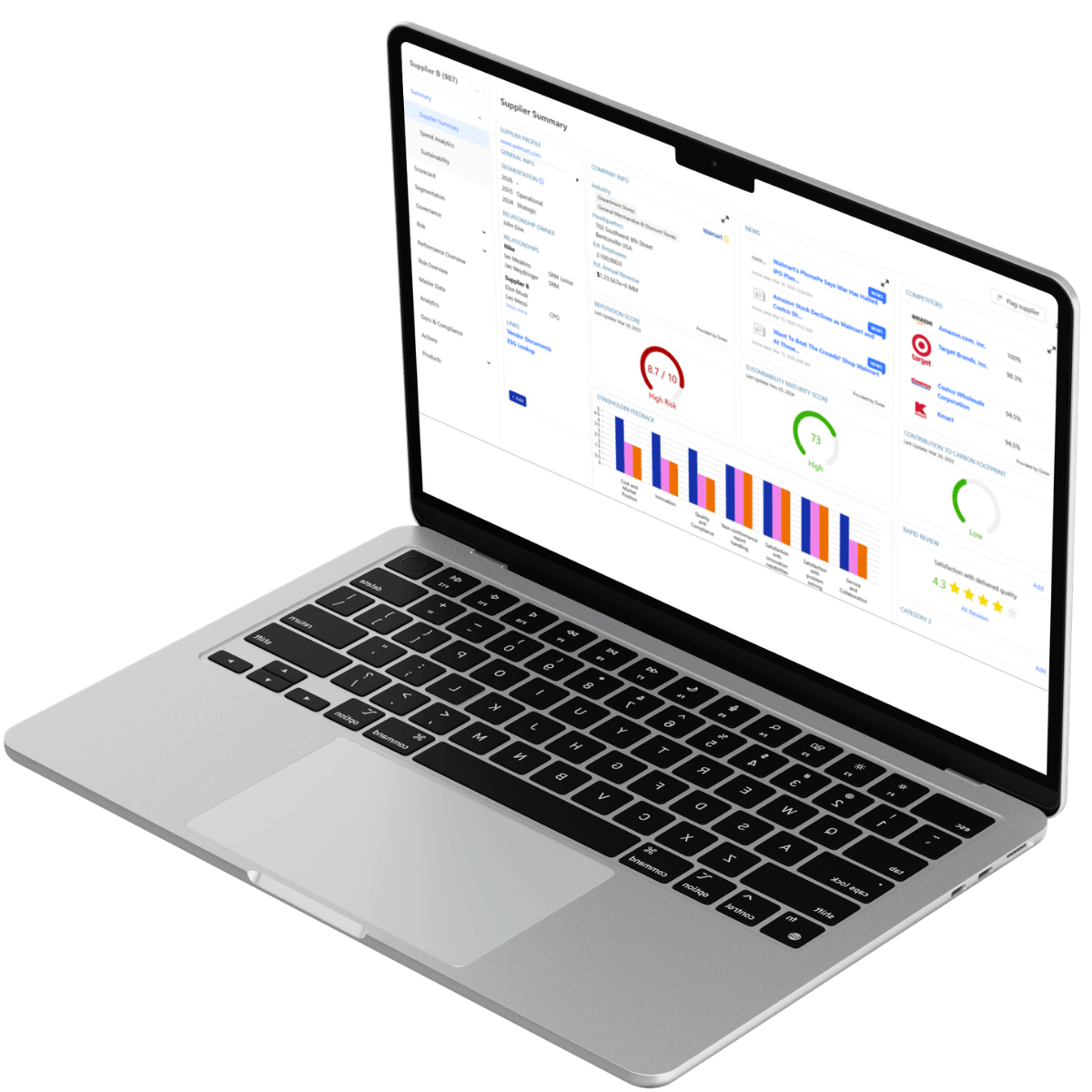
Centralized Master Data & Self-Service Portals
Governance begins with structured data.
Supplier master data management creates a Single Source of Truth by centralizing:
- Bank details
- Tax documentation
- Compliance certificates
- Supplier classification data
Through a transparent and structured supplier portal, suppliers upload and maintain their own documentation. This reduces manual entry errors and improves data validation accuracy.
To eliminate data entry errors during the initial engagement, utilize Supplier Onboarding Software that forces suppliers to register and self-validate their documentation.
Clean master data is the foundation to any modern supply chain management cloud software environment.
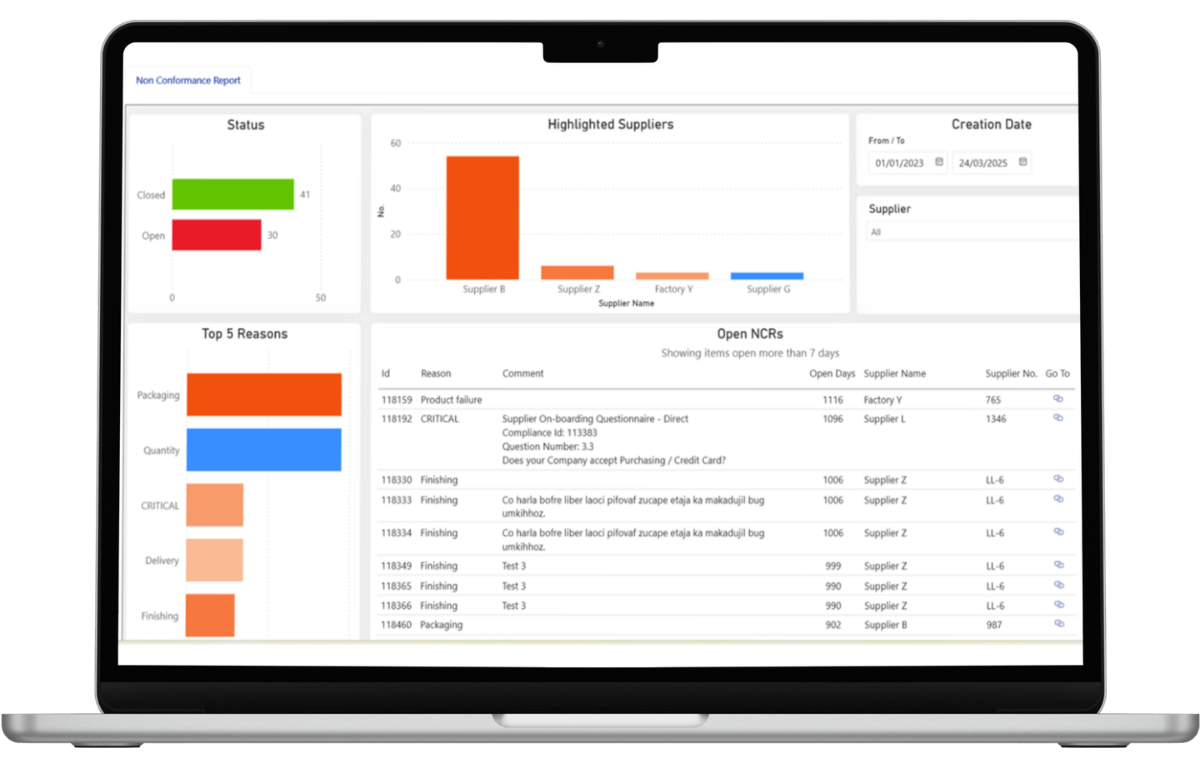
Non-Conformance Reporting (The “Fix-It” Engine)
This module differentiates governance systems from static databases.
NCR software governs the active handling of deviations such as:
- Damaged goods
- Incorrect quantities
- Specification failures
- Labelling issues
The system enforces:
- Root Cause Analysis (RCA)
- Corrective and Preventive Actions (CAPA)
- Escalation timelines
- Resolution validation
Effective quality control relies on detailed Supplier Quality Management workflows to track RCA and CAPA enforcement. This converts deviation handling from informal email exchanges into structured, enforceable operational governance that automatically can be an input to the supplier performance scorecard.
Optimized for Regulated Manufacturing: Pharma, Food, and Industrial Supply Chains
These supplier management software solutions are engineered specifically for regulated industries.
 Manufacturing
Manufacturing Food & Beverage
Food & Beverage Pharmaceutical
PharmaceuticalFrom Data Collection to Automated Scorecarding
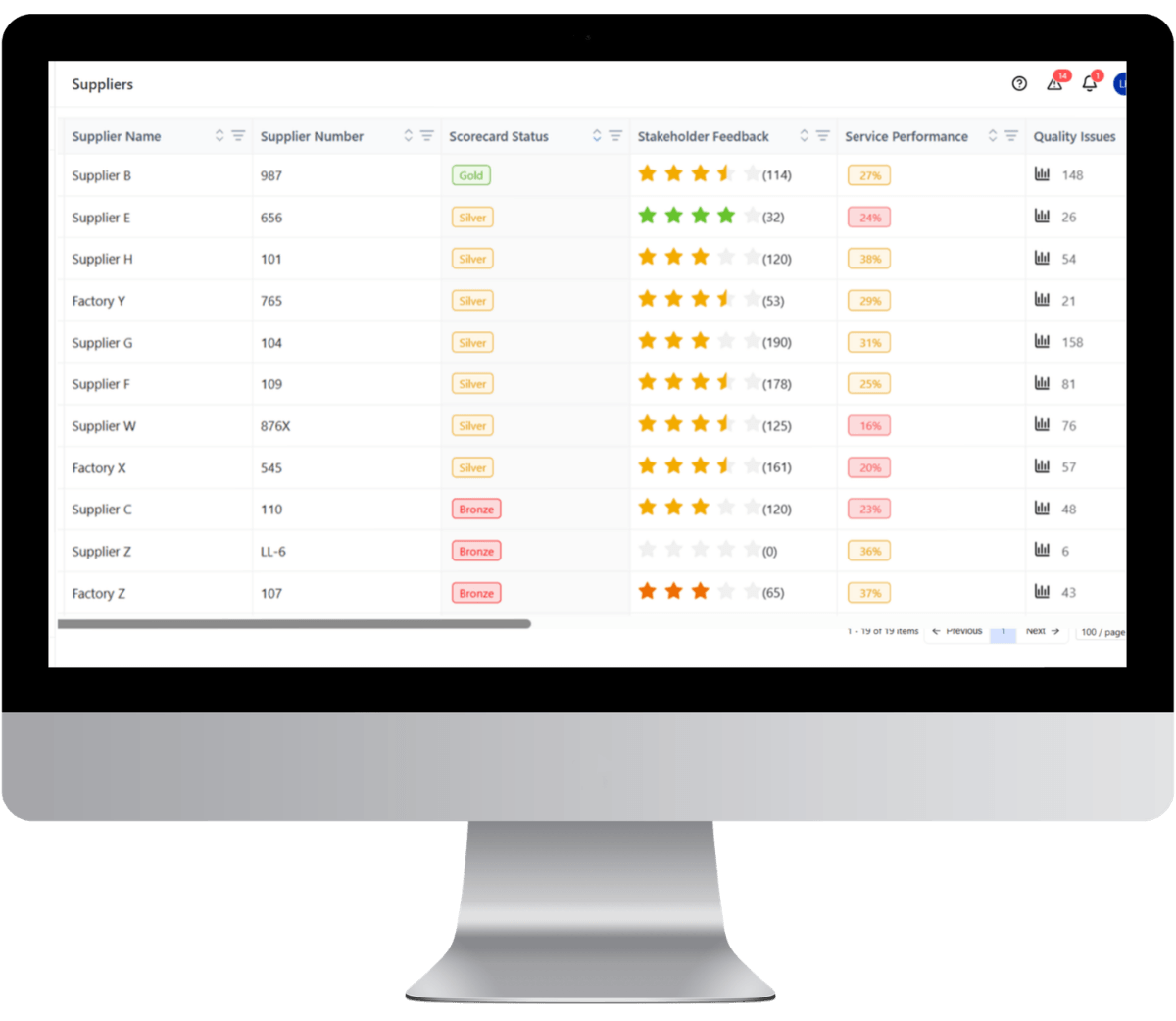
You cannot score what you cannot measure and see.
Centralized supplier master data management forms the baseline for performance measurement inside advanced supply chain management software
Validated compliance data enables
- Fact-Based Negotiation
- Objective supplier evaluation
- Risk-based segmentation
Automated scorecard triggers
- Certificate expiry detection
- Compliance document lapse alerts
- Quality deviation escalation
- Declining performance flags
What governance unlocks
Transactional systems store data. Governance systems enforce accountability. The shift from passive ERP records to an active System of Engagement is what separates reactive supply chains from resilient ones.
Testimonials

"LeanLinking is a game changer for working with suppliers. For the first time we can close the supplier management cycle from strategy through governance to performance managements and supplier development."

"Our team loves easy to use interface; with Relations, we have managed to cut non-compliance resolution time by half, and migrated interactions with our vendors almost fully to LeanLinking."

Take Your Supplier Quality from Reactive Control to Zero-Defect Enforcement
LeanLinking replaces reactive quality control with structured workflows that enforce defect resolution, validate corrective actions, and maintain audit-ready traceability across the supply chain.
Supplier Management FAQs
What is the difference between Supplier Management Software and an ERP?
While ERPs focus on the financial transaction (invoice and PO), Supplier Management Software governs the operational and strategic data exchange (compliance, quality, performance and more) related to suppliers.
- ERPs operate as passive Systems of Record.
- Supplier Management Software functions as a proactive System of Engagement.
ERPs record transactions. An advanced Vendor Management System triggers workflows when:
- A certificate expires
- A compliance document lapses
- A quality deviation occurs
- Supplier performance is declining
This distinction becomes critical in any supply chain management software comparison. Transactional systems store data. Governance systems enforce accountability.
To understand why legacy systems fail at supplier collaboration, read our comparison on LeanLinking vs. SAP Ariba.
Real Stories, Real Results
E-Book · 4 min
Case Study · 4 min




