Supplier Quality Management Software:
The Zero-Defect Command Center for Regulated Supply Chains
LeanLinking’s Supplier Quality Management Software centralizes supplier quality oversight by digitizing defect resolution, enforcing corrective actions, and maintaining full audit-ready traceability across regulated supply chains.
Structured Governance for Supplier Quality Management
LeanLinking’s Supplier Quality Management Software is a collaborative governance system that digitizes Non-Conformance Reports (NCRs), mandates Root Cause Analysis (RCA), enforces Supplier Corrective Action Requests (SCARs), and centralizes supplier audit data to prevent quality failures from reaching production. It functions as an ongoing enforcement engine in regulated supply chains where unresolved deviations result in operational disruption, regulatory exposure, and commercial loss.
Structured Quality Issue Management
LeanLinking digitizes supplier quality workflows by capturing defects, enforcing root cause investigation, and managing corrective actions within a centralized governance system.
Automated Quality Enforcement
The platform acts as an automated quality firewall that blocks unresolved defects, validates corrective actions before closure, and ensures issues cannot be informally resolved or ignored.
Continuous Compliance Governance
Quality governance cannot be periodic. LeanLinking ensures compliance oversight is continuous, documented, and enforceable across the supplier base.
The Failure of Email Quality Control: Managing Defects in the Dark
Many organizations still manage supplier quality through Outlook threads, Excel trackers, and shared drives. This fragmented structure creates blind spots across the supply chain where defects are reported informally, corrective actions are discussed through email attachments, and critical documentation becomes scattered across inboxes rather than controlled systems.
Over time, this approach distributes responsibility without enforcing accountability.
No Enforced Resolution Discipline
Suppliers respond selectively because there is no structured requirement to submit documented Root Cause Analysis before a case is closed. Corrective actions may be promised but are not consistently validated.
No Centralized Defect History
Recurring issues cannot be analyzed systematically because prior NCRs and quality cases remain buried across emails, attachments, and spreadsheets rather than recorded in a centralized defect history.

No Audit-Grade Traceability
When documentation is fragmented, organizations struggle to demonstrate who authorized a deviation, when corrective action was approved, or whether preventive measures were implemented during inspections.
Core Pillars of Automated Quality Control: NCRS, SCARS, and Digital Audits
Zero-defect governance requires structured resolution artifacts that eliminate ambiguity and enforce discipline.
Automated NCR Management:
The “Fix-It” Engine
LeanLinking’s automated NCR management converts defect detection into structured resolution.
When a quality deviation is identified, the system:
- Generates a Non-Conformance Report
- Assigns it to the supplier
- Applies deadline enforcement
- Tracks response status in real time
- Flags overdue cases with visible escalation indicators
LeanLinking’s Supplier Quality Management platform prevents suppliers from closing an NCR without submitting a documented Root Cause Analysis. Corrective actions must be measurable and timely. Preventive actions can be mandated to eliminate systemic recurrence.
Every interaction is logged. Every status change is recorded. Every approval is auditable.
Scandi Standard deployed structured NCR workflows across its factories and reduced non-compliance resolution time by 50 percent. This reduction was not driven by faster email response; it was achieved through enforced workflow discipline and centralized visibility.
LeanLinking’s system does not store defects, it drives resolution.
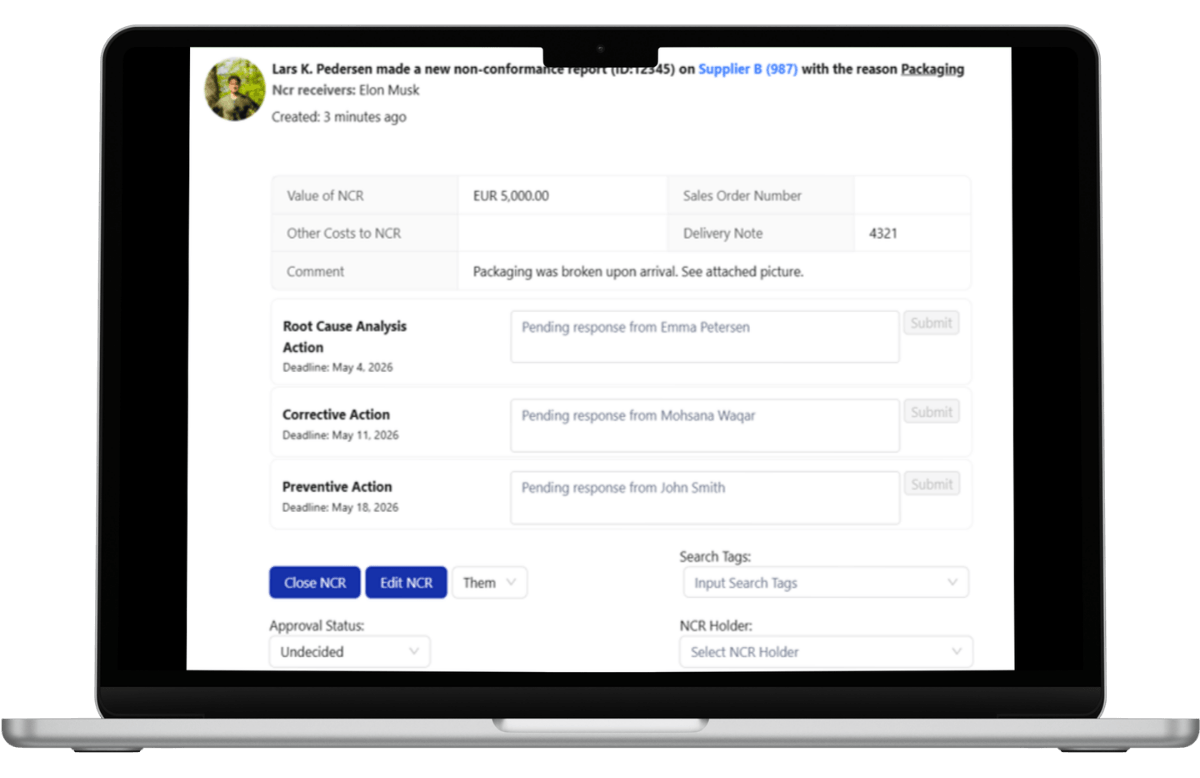
Collaborative Problem Solving with Full Audit Traceability
Supplier quality enforcement requires structured collaboration.
Within LeanLinking’s centralized portal, suppliers and Quality Managers communicate directly on the defect record. Comments, attachments, Route Cause Analysis, and approval decisions are captured within the NCR case history.
This eliminates:
- Parallel email conversations
- Version confusion
- Undocumented agreements
- Informal approvals
Every resolution step becomes part of a permanent audit trail.
Structured collaboration does not end with case closure. With LeanLinking, NCR activity feeds directly into Supplier Performance Scorecards, automatically updating quality KPIs such as defect trends, average resolution time and corrective action responsiveness.
This ensures that supplier quality performance is continuously reflected in scorecard evaluations rather than reviewed retrospectively. Defect history becomes a measurable governance input that influences supplier ratings, segmentation decisions, and future commercial discussions.
Quality collaboration therefore extends beyond issue resolution. It directly impacts how supplier performance is quantified, compared, and enforced across the supplier base.
Digital Supplier Audits and Threshold-Based Enforcement
Quality governance extends beyond reactive defect control; it requires proactive audit validation.
LeanLinking’s digital audit features distribute structured self-assessment questionnaires to suppliers, completion status is tracked in real time, and responses that fall below defined compliance thresholds trigger mandatory follow-up workflows.
Audit responses can be linked to supplier risk categories, automatically increasing oversight where compliance deviations occur.
This transforms audits from static annual exercises into dynamic compliance enforcement mechanisms.
Good Food Group (internal linking to Good Food case study) uses LeanLinking as the technological backbone for food safety governance. Digital audit enforcement ensures ingredient suppliers meet strict quality and certification standards before entering production.
With LeanLinking audit readiness is continuous and compliance is measurable.
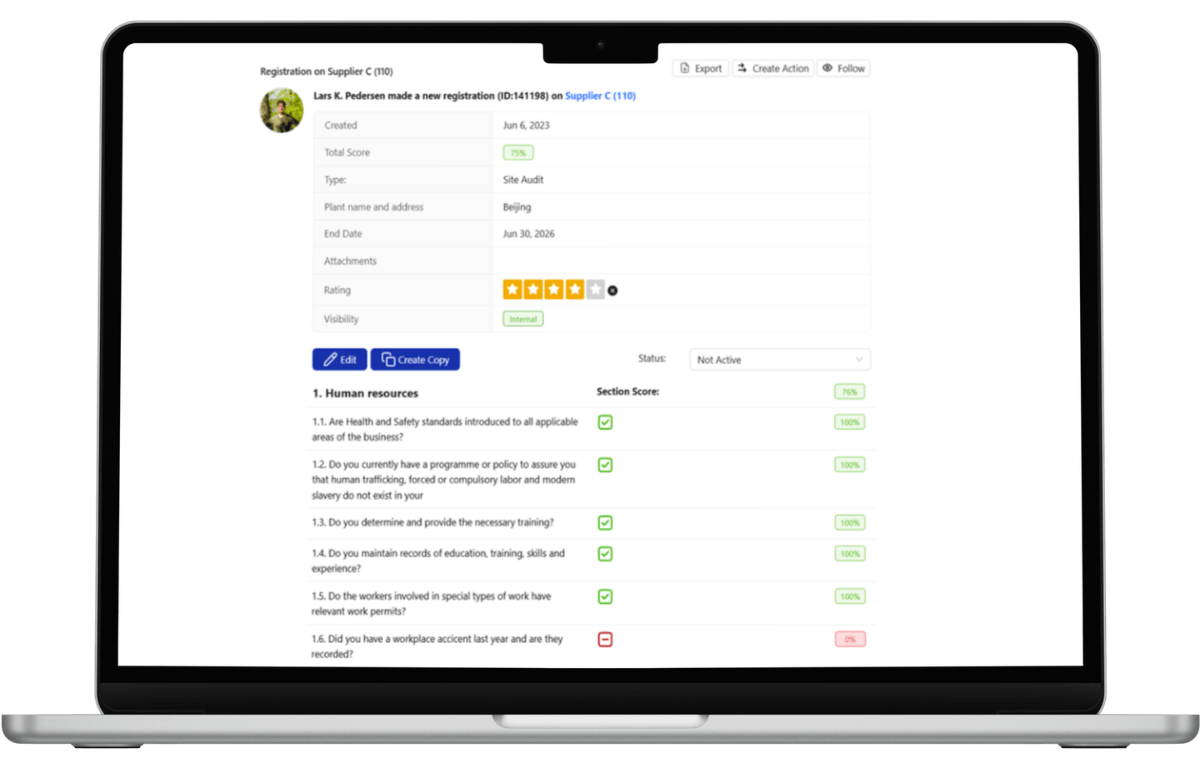
Built for Regulated and Physical Supply Chains
Zero-defect enforcement is mission-critical in physical supply chains where quality failures disrupt production lines and trigger regulatory action.
Across these industries, quality governance is not optional, it is infrastructure.
 Manufacturing
Manufacturing Food & Beverage
Food & Beverage Pharmaceutical
PharmaceuticalTurning Quality Data into Commercial Leverage
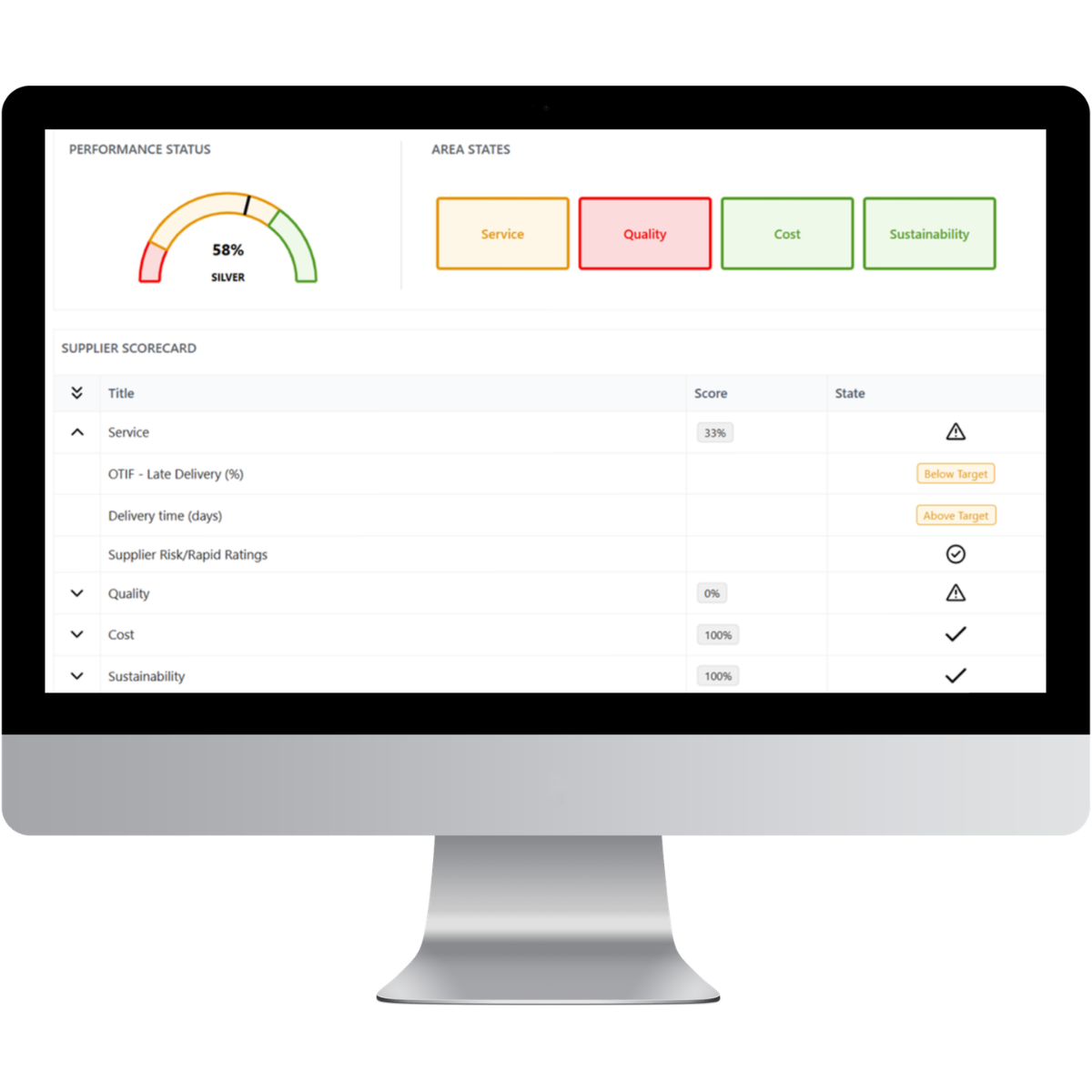
You cannot negotiate what you cannot measure. LeanLinking transforms supplier quality data into structured performance intelligence that procurement and quality teams can use to strengthen supplier governance and commercial negotiations.
Defect Data Transparency
Every defect case generates measurable operational data that provides objective insight into supplier performance.
- Defect frequency
- Response time
- Recurrence rate
- Corrective action effectiveness
- Cost-of-poor-quality impact
Fact-based supplier negotiations
With LeanLinking, procurement teams replace subjective arguments with verified operational performance data. Documented defect history and corrective action outcomes provide measurable evidence during supplier negotiations and contract discussions.
Operational data strengthens supplier governance
When manufacturers integrate defect analytics into procurement strategies, supplier performance becomes a measurable commercial variable. This allows organizations to hold suppliers accountable and make sourcing decisions based on validated operational performance.
Testimonials

"LeanLinking is a game changer for working with suppliers. For the first time we can close the supplier management cycle from strategy through governance to performance managements and supplier development."

"Our team loves easy to use interface; with Relations, we have managed to cut non-compliance resolution time by half, and migrated interactions with our vendors almost fully to LeanLinking."

Take Your Supplier Quality from Reactive Control to Zero-Defect Enforcement
LeanLinking replaces reactive quality control with structured workflows that enforce defect resolution, validate corrective actions, and maintain audit-ready traceability across the supply chain.
Supplier Quality Management FAQs
Why do companies need a quality governance system in addition to ERP data?
ERP systems such as SAP and Oracle are designed to record internal transactions. They track returns, goods receipts, and inventory adjustments. However, they do not enforce supplier accountability at the root cause level.
When a defect is logged in ERP, the system captures the operational impact. It does not mandate supplier corrective action.
ERP modules are reactive. However, quality governance needs to be proactive.
LeanLinking’s dedicated Supplier Quality Management portal operates above the ERP to enforce collaborative workflows between the organization and its suppliers.
When a defect is detected:
- An automated NCR is issued directly to the supplier
- The supplier is required to submit documented Root Cause Analysis
- Corrective and preventive actions must be defined
- The Quality Manager reviews and either approves or rejects the submission
- The case remains open until validated resolution is achieved
The system blocks closure until resolution criteria are satisfied.
This enforcement architecture ensures that every defect progresses through a controlled lifecycle rather than disappearing into email threads.
Organizations comparing ERP-native quality tools with dedicated governance platforms often evaluate this distinction in structured assessments such as LeanLinking vs. SAP Ariba (internal linking to article), where the enforcement depth and supplier collaboration layer become critical differentiators.
Real Stories, Real Results
E-Book · 4 min
Case Study · 4 min




