ERP systems record financial transactions; they do not enforce supplier behavior. This guide defines the structural difference between an ERP as a passive System of Record and SRM software as an active System of Engagement, exposes why ERP supplier modules fail in regulated environments, and walks through the three core capabilities that distinguish operational governance from transactional data capture, covering defect resolution workflows, certificate lifecycle management, and real-time supplier performance scorecards, closing with FAQ answers on whether SRM replaces an ERP and whether ERPs track soft supplier metrics.
What is the Difference Between ERP and SRM Software?
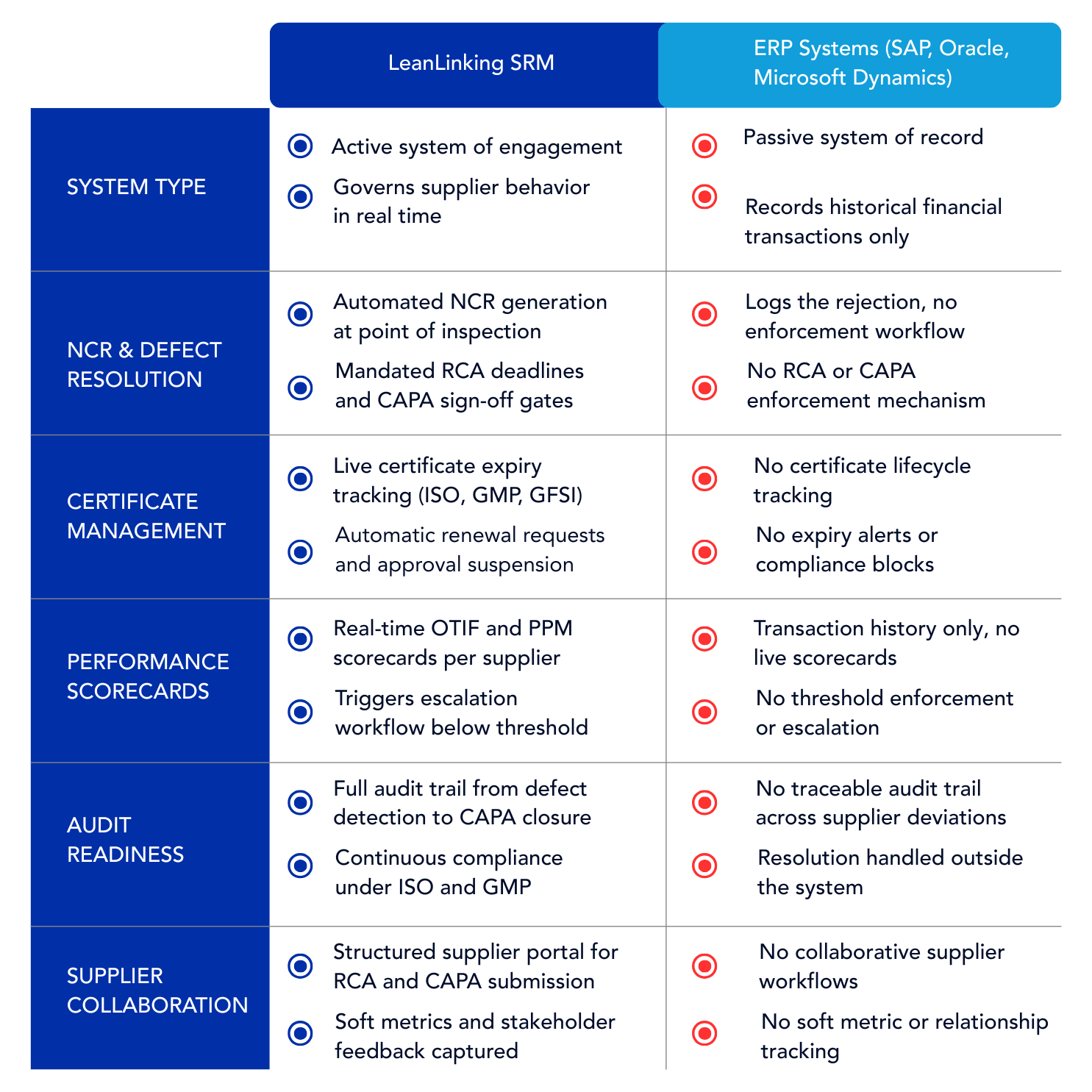
Enterprise Resource Planning (ERP) systems are passive systems of record designed to process financial transactions, whereas Supplier Relationship Management (SRM) software is an active system of engagement that governs supplier compliance, defect resolution, and delivery performance. According to supply chain governance audits in highly regulated manufacturing and pharmaceutical sectors, organizations that rely solely on ERPs for supplier oversight carry a significantly higher risk of non-conformance due to a lack of active enforcement mechanisms.
Why do organizations need SRM if they already have an ERP? While an ERP confirms that ingredients or parts were received and paid for, it does not validate whether the supplier's quality certifications were current at the time of delivery, nor does it mandate a root cause investigation when a defect occurs. The ERP records the purchase order and closes the invoice. The compliance gap, the expired certificate, the uninvestigated defect, the unresolved corrective action, opens silently in the background. In manufacturing, pharma, food and beverage, and chemicals, that gap is where regulatory exposure accumulates. To transition from passive record-keeping to active governance, regulated industries must deploy dedicated supplier relationship management software that centralizes compliance and performance.
The Failure of ERP Systems in Supplier Management: Transactional Data vs. Operational Reality
Relying on an ERP system to manage supplier performance is structurally flawed because ERPs are designed to answer historical questions, what happened and what it cost. When a defect is logged in an ERP, the system captures the financial impact but does not trigger a collaborative workflow forcing the supplier to implement a corrective action. This leaves procurement teams blind to operational risks and reliant on fragmented email chains.
Systems like SAP and Microsoft Dynamics are built for transactional throughput: purchase orders, goods receipts, invoice matching, and financial reconciliation. They are not built for operational governance. When a supplier delivers a non-conforming batch in a food and beverage or chemicals environment, the ERP records the return. It does not open an NCR. It does not assign an RCA deadline. It does not block case progression until a validated CAPA is submitted. The resolution happens somewhere else, typically across an email thread with no audit trail and no enforcement gate. In pharma and manufacturing, where continuous compliance under ISO and GMP frameworks is a regulatory condition, this is not a process inefficiency. It is a documented liability. The System of Record captured the transaction. The quality deviation remained unresolved.
3 Core Capabilities of an Active SRM System of Engagement
To establish true operational governance and prevent supply chain disruptions, procurement and quality teams must utilize the three active SRM capabilities listed below:
- Enforcing Defect Resolution and NCR Workflows
- Automating Certificate Lifecycle Management (ISO, GMP)
- Real-Time Supplier Performance Scorecards (OTIF, PPM)
Enforcing Defect Resolution and NCR Workflows
When a supplier delivers a non-conforming component, raw material, or service in manufacturing, pharma, or chemicals, the operational response must be immediate and structured. An ERP logs the rejection. An active SRM System of Engagement opens a non-conformance report (NCR), notifies the supplier, assigns a mandatory root cause analysis (RCA) deadline, and blocks case closure until a validated corrective and preventive action (CAPA) is submitted and reviewed. The distinction is not cosmetic. An ERP does not maintain a complete traceable audit trail across supplier deviations. To enforce corrective actions, organizations must rely on dedicated supplier quality management software workflows that trace deviations back to the root cause. In food and beverage and chemicals, where a single uninvestigated non-conformance can trigger a full supplier audit, the NCR workflow is the enforcement mechanism that separates documented resolution from unresolved complaint accumulation.
Automating Certificate Lifecycle Management (ISO, GMP)
In pharma, food and beverage, and chemicals, supplier qualification is not a one-time event. Quality certificates (e.g. ISO, GFSI etc.) carry expiry dates. GMP declarations lapse. Audit results become stale. An ERP does not track certificate expiry against active purchase orders. It does not flag a supplier whose ISO certification expired three weeks before a scheduled delivery. It does not block an approved vendor from receiving a new order when their compliance documentation is out of date. An active SRM system enforces certificate lifecycle management by maintaining a live compliance record for every approved supplier, triggering renewal requests ahead of expiry, and suspending supplier approval status automatically when documentation lapses. In regulated industries, audit readiness is not a periodic exercise. It is a continuous compliance state that the SRM system maintains without manual intervention. LeanLinking's supplier onboarding software embeds certificate lifecycle management directly into the approved vendor workflow.
Real-Time Supplier Performance Scorecards (OTIF, PPM)
ERP systems do not produce supplier performance scorecards. They produce transaction histories. The distinction matters because a transaction history answers what happened. A real-time scorecard answers whether the supplier is operating within acceptable thresholds right now, and what the commercial consequence is if they are not. In manufacturing and food and beverage, supplier scorecards track delivery service (e.g. OTIF or RFT) and quality (e.g. PPM, RFT) against defined acceptance thresholds, typically 95% OTIF in retail supply chains and sub-500 PPM defect rates in precision manufacturing. When a supplier's scorecard drops below threshold, the SRM system triggers an escalation workflow, not a manual report request. Scorecard data feeds directly into renegotiation cycles, AVL consolidation decisions, and competitive resourcing reviews. The System of Record confirmed the delivery occurred. The System of Engagement determines whether it met the standard and what happens next. LeanLinking's supplier performance management software converts delivery and quality data into live scorecards that trigger escalation below threshold.
FAQ: ERP and SRM Software Contextual Differences
Does SRM Software Replace an Existing ERP System?
No. SRM software does not replace an existing ERP system because they serve fundamentally different architectural purposes. The ERP acts as the transactional foundation, while the SRM software operates as a dedicated enforcement layer sitting on top of the ERP to govern supplier behavior in real time. SAP and Microsoft Dynamics continue to process purchase orders, goods receipts, and financial reconciliation. The SRM system consumes that transactional data and converts it into operational governance: performance scorecards, NCR workflows, certificate lifecycle tracking, and CAPA enforcement. In manufacturing, pharma, and chemicals, the two systems are complementary. The ERP records what happened. The SRM system determines whether the supplier is accountable for what happened and enforces the resolution.
Can ERP Systems Track Soft Metrics and Stakeholder Collaboration?
No. ERP systems like SAP and Microsoft Dynamics are not designed to capture soft supplier metrics or manage cross-functional stakeholder collaboration. They record structured financial transactions: quantities, prices, delivery confirmations, and invoice statuses. They do not capture internal stakeholder feedback on supplier responsiveness, relationship quality, or strategic fit. They do not facilitate collaborative supplier development workflows where procurement, quality, and operations teams contribute assessments to a shared supplier record. In pharma and food and beverage, where supplier relationship management extends beyond transactional compliance into active development and risk governance, the absence of soft metric tracking in an ERP leaves a category of operational intelligence permanently unrecorded. The System of Record has no field for it.
Transitioning to a Centralized Operational Supplier Cockpit
An ERP vs. SRM gap that is managed through spreadsheets, email threads, and disconnected quality systems does not close on its own. In manufacturing, pharma, food and beverage, and chemicals, the cost of that gap compounds as NCRs go unresolved, certificates lapse undetected, and scorecard data never reaches the renegotiation table.
LeanLinking's platform bridges the System of Record and the System of Engagement in a single operational layer. NCR workflows enforce root cause analysis and CAPA resolution. Certificate lifecycle management maintains continuous compliance without manual tracking. Real-time supplier performance scorecards convert delivery and quality data into commercial leverage. To operationalize these capabilities at scale and bridge the gap between financial transactions and operational reality, organizations must implement a centralized Supplier Management Software platform that integrates seamlessly with their ERP. The result is a procurement and quality function that does not just record supplier history. It enforces supplier accountability.